

A research team led by Prof Yu Zhengquan from the School of Life Sciences at Zhengzhou University (ZZU) has uncovered a key mechanism linking sleep disturbance to intestinal dysfunction. The study, published in journal Cell Stem Cell, demonstrates that sleep loss triggers a neural pathway that directly impairs the regenerative capacity of intestinal stem cells.
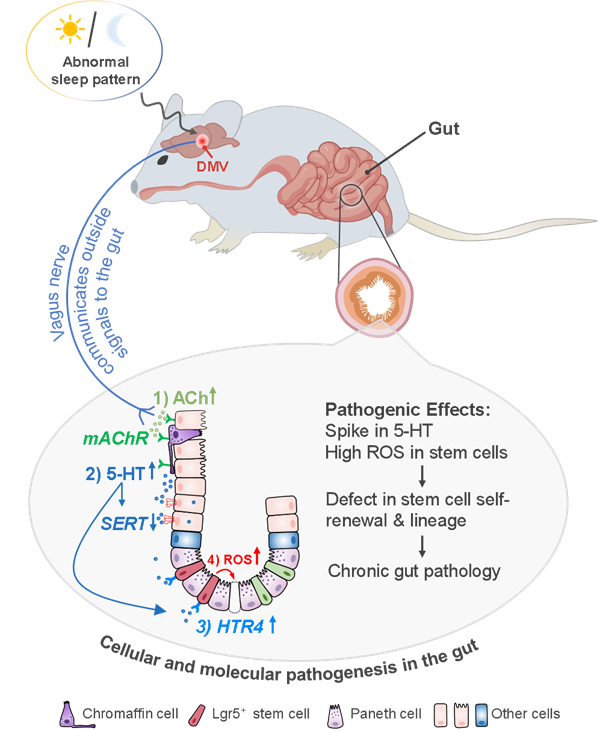
ZZU study reveals how sleep disruption harms gut health via neural pathway. [Photo/zzu.edu.cn]
Using mouse models, the team found that just two days of sleep deprivation reduced intestinal stem cell numbers by half. The damage is mediated by a specific brain region — the dorsal motor nucleus of the vagus (DMV) — which becomes abnormally activated during sleep loss. This activates the vagus nerve to release acetylcholine in the gut, prompting enterochromaffin cells to produce excessive serotonin (5-HT). High local serotonin levels then activate receptors on intestinal stem cells, inducing oxidative stress and impairing their ability to self-renew and repair.
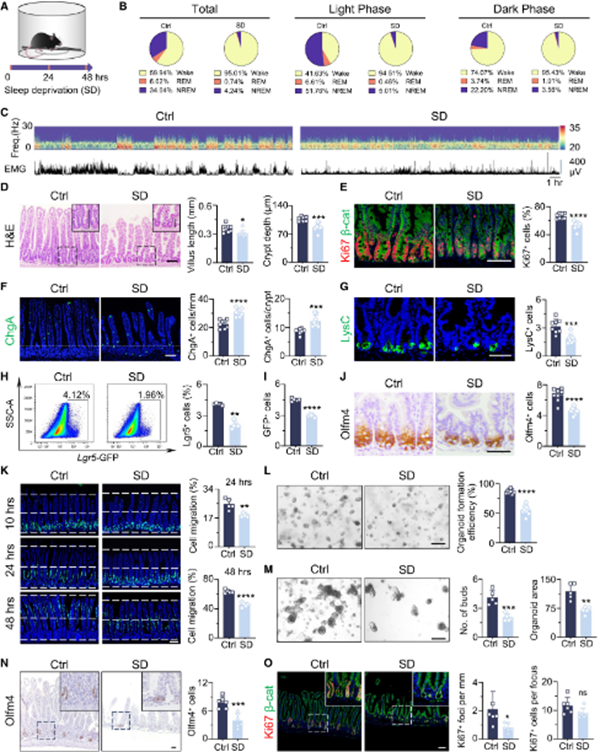
Short-term SD significantly impaired ISC functions and disrupted intestinal epithelial cell turnover. [Photo/zzu.edu.cn]
While serotonin is well-known as a brain neurotransmitter, approximately 95 percent of the body's serotonin is actually produced in the gut. The study highlights its critical role as a peripheral signaling molecule linking brain states to intestinal health.
Encouragingly, the researchers identified several protective interventions that can block this damage pathway, including drugs that inhibit muscarinic receptors or serotonin receptors, as well as the antioxidant vitamin C.
The findings provide direct evidence for how sleep disruption affects the "brain-gut axis" and may have broader implications for understanding how sleep disorders contribute to multi-organ dysfunction.
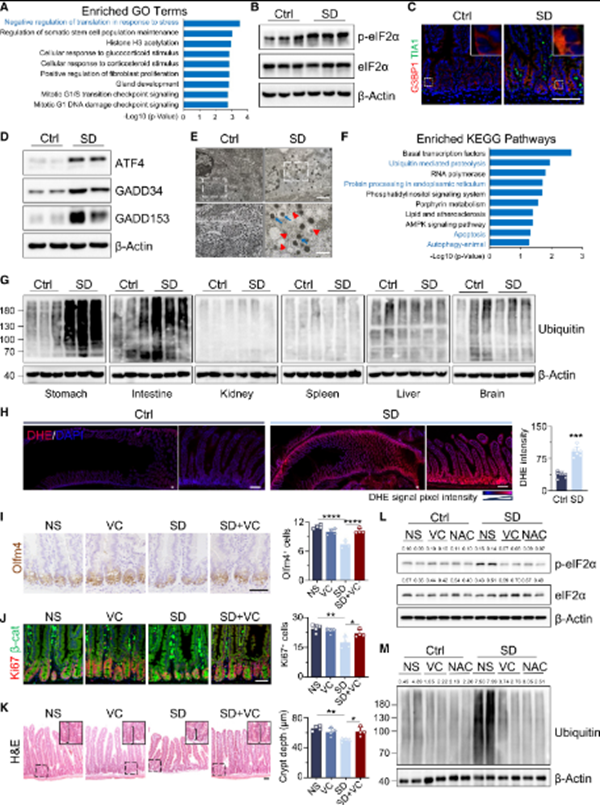
Short-term SD compromises ISCs by activating the translation stress response. [Photo/zzu.edu.cn]