

A research team from the College of Chemistry at Zhengzhou University (ZZU), in collaboration with Professor Stephen T Liddle's group at the University of Manchester, has successfully constructed inverse-sandwich complexes featuring an all-bismuth three-membered ring stabilized by dual uranium or thorium centers. The work is published in Nature Chemistry.
ZZU chemists achieve a breakthrough in actinide chemistry. [Photo/zzu.edu.cn]
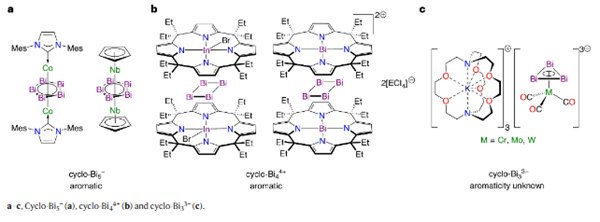
Aromaticity is a fundamental concept in organic chemistry, exemplified by benzene and the cyclopropenyl cation. However, whether the Huckel model applies to heavier elements remains debated. The cyclo-Bi3 ligand is the heaviest analogue of the cyclopropenyl group, but planar Bi3 rings are difficult to isolate due to bismuth's tendency to form cluster structures.
ZZU chemists achieve a breakthrough in actinide chemistry. [Photo/zzu.edu.cn]
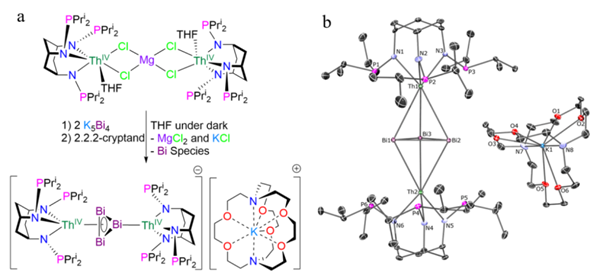
To overcome this challenge, the team designed a new multidentate cyclohexanetriamine ligand and reacted thorium chloride with K5Bi4 as a cyclo-[Bi3]3- transfer reagent under light-protected conditions. This yielded pure crystalline products of Th2Bi3 complexes containing a planar cyclo-[Bi3]3- ring in an inverse-sandwich configuration.
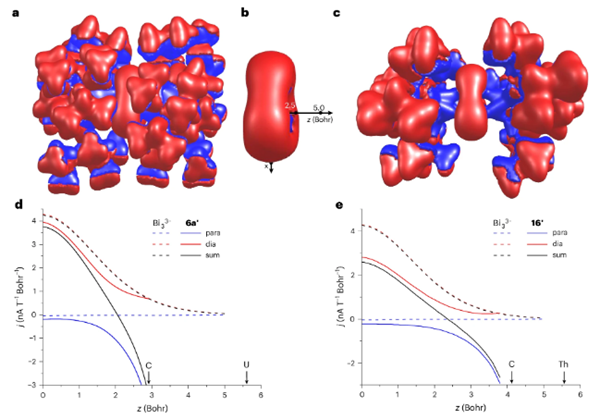
This finding provides the first clear evidence of all-metal aromaticity in a heavy p-block ring system and broadens our understanding of aromaticity beyond traditional organic frameworks.
ZZU chemists achieve a breakthrough in actinide chemistry. [Photo/zzu.edu.cn]